Our Technology
Developing unique proprietary RNA therapies for cardiovascular disease delivered systemically, including pulmonary hypertension and atherosclerosis
THE DISCOVERY
In 2014, the ground breaking discovery of vascular specific LNPs was published in Nature by the pioneers of RNAi/LNP technology, profs Robert Langer and Dan Anderson from MIT. Their unique LNP platform was able to specifically transfect vasculature (i.e. vascular endothelium) following a simple iv bolus, without any transgenic expression in the liver (i.e. hepatocytes; Dahlman et al, Nature Nanotechnology, 2014).
VasoRx received an exclusive global license from MIT to use this technology and was able to innovate this breakthrough lipid nanoparticle platform which allows targeting of the vasculature, healthy, as well as diseased vasculature. The platform is able to transduce with equal efficacy the endothelial lining of large vessels, as well as microvasculature (arteries, arterioles and capillaries). The technology has been applied to a wide spectrum of disease, varying from cardiovascular disease, inflammatory disease, auto immune disease, as well as oncology to name a few.
Dysfunctional endothelial cells of the vascular lining contribute to more disease than any other tissue in the body. MicroRNAs (miRNAs) and small interfering RNAs (siRNAs) have the potential to help study and treat endothelial cells by durably silencing multiple genes simultaneously, but efficient RNA delivery has so far remained challenging. Our unique VasoRx polymeric nanoparticles made of low molecular weight polyamines and lipids can deliver RNAi and mRNAs to endothelial cells with extreme high efficiency, thereby facilitating the simultaneous modification of expression of multiple endothelial genes in vivo.
Based on research at Yale University and MIT, endothelial-to-mesenchymal transition has been shown to be a biological process that underlies multiple diseases, and is responsible for the ongoing chronic vascular inflammation, fibrosis formation, and adaptive (vascular) remodeling, leading to further deterioration of the disease process and progressive symptoms. By blocking the endothelial-to-mesenchymal transition in the vascular lining of endothelial cells, VasoRx has been able, for the first time, to reverse the vascular changes and symptoms seen in cardiovascular and cardiopulmonary disease models, like pulmonary hypertension and atherosclerosis models.
By a simple iv infusion of the unique polymeric LNP carrying a specific RNAi for 4 weeks, the vascular changes and elevated pressure in pulmonary vascular bed were significantly alleviated, if not normalized in a pulmonary hypertension disease model.
Likewise, in a high-fat atherosclerosis disease model, treatment with the polymeric LNP carrying a specific RNAi, the volume of pre-existing atherosclerotic plaques was significantly reduced by 60% with a marked reduction of local inflammatory cells (foam cells) and volume of the necrotic core. These anti-inflammatory effects of our LNPs in atherosclerosis suggest that the LNP therapy is able to lead to plaque regression and stabilize the so-called fatty or ‘vulnerable’ atherosclerotic plaques, into stable (fibrotic) atherosclerotic plaques.
The unique LNP-RNAi approach safely promotes
Selective silencing of the targeted mRNA in endothelial cells for the period of week, whereas non-endothelial cells were unaffected
Reversal of vascular remodeling, as seen in pulmonary hypertension and atherosclerosis models
Reversal of the elevated pulmonary hypertension in pulmonary hypertension disease models
Reversal of chronic vascular inflammation (reduction of foam cells and inflammatory cells)
Reversal of atherosclerotic plaque volume
Reversal of vulnerable atherosclerotic plaque phenotype into stable fibrotic atherosclerotic plaque phenotype
RNAi Therapy in Cardiovascular Disease
OUR PRODUCT
VSR03
Systemic Delivery of VSR Polymeric Nanoparticles to Revert Pulmonary Hypertension and Vascular Remodeling in Pulmonary Arterial Hypertension Patients
We are developing an IV formulation of LNPs that target and specifically reprogram lung vascular cells (endothelial cells) to modulate the endothelial-to-mesenchymal transition that drives vascular changes and chronic inflammation in pulmonary hypertension. The therapy can be given every 30 days at an outpatient clinic.
Different formulations of the API to enable other routes of delivery are being developed.
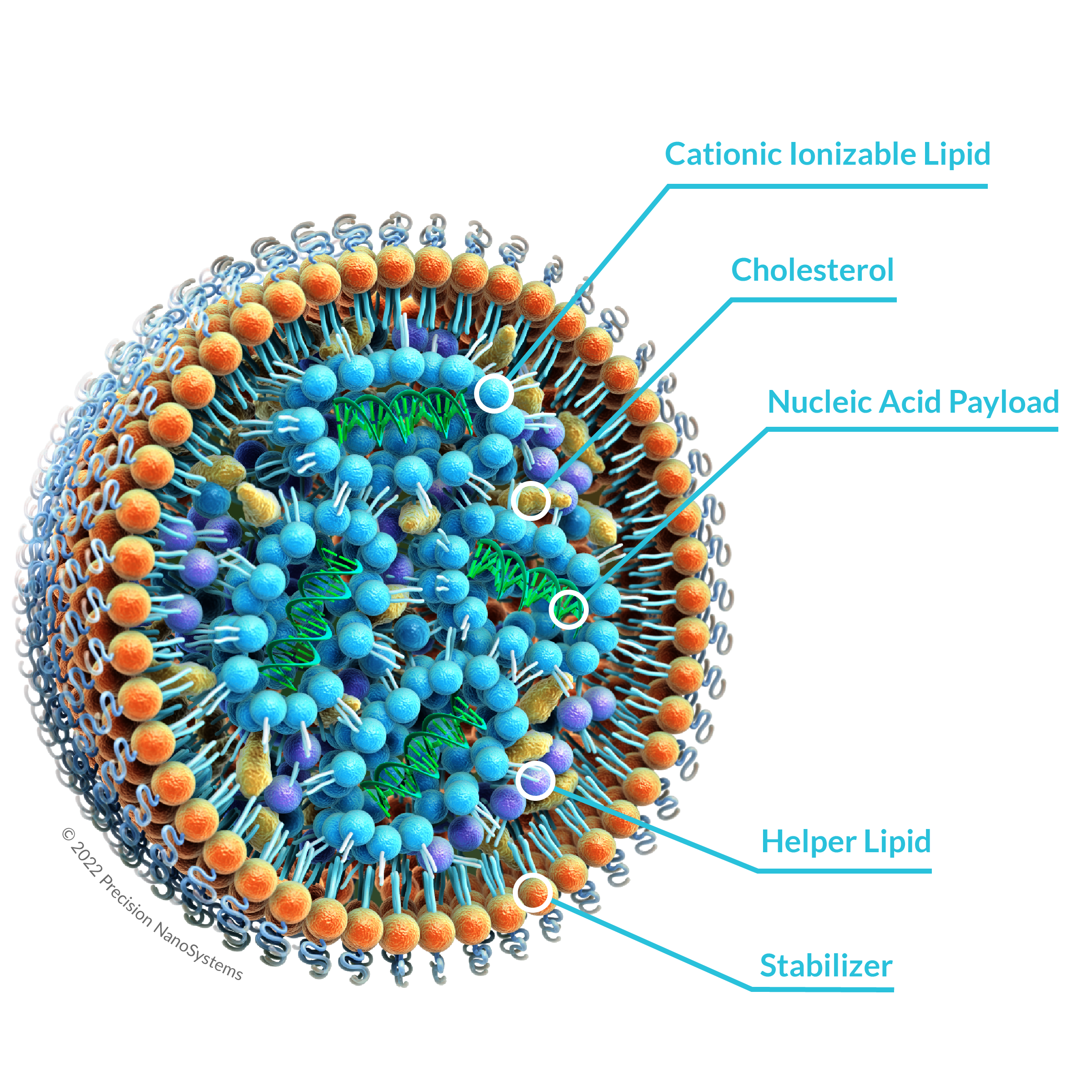
VSR05
Systemic Delivery of VSR Polymeric Nanoparticles to induce regression and stabilization of fatty atherosclerotic plaques (vulnerable atherosclerotic plaques) in cardiovascular patients
We are developing an IV formulation of LNPs that target and specifically reprogram coronary vascular cells of the heart, and carotid vascular (endothelial) cells to modulate the mechanisms that drives chronic inflammation and vascular obstruction (clogging) in patients with coronary artery disease and cerebrovascular disease.
The therapy can be given every 30 days at an outpatient clinic.
Different formulations of the API to enable other routes of delivery are being developed.
If you are interested in learning more about the science behind our technology, check out our selected literature

The unique elements of VasoRx’s Therapeutic approach:
INTELLECTUAL PROPERTY
Issued Patents & Highlights of this patent family:
Composition of Matter, Pharmaceutical Compositions and Uses of 7C1-based non-cationic lipoprotein nanoparticles that target vasculature/ endothelial cells issued in the US, Japan and Australia
Composition of Matter, Pharmaceutical Compositions and Uses of targeting the TGFbeta pathway to modulate the endothelial-to-mesenchymal transition to inhibit vascular remodeling and chronic vascular inflammation as seen in primary and secondary pulmonary (arterial) hypertension, atherosclerosis and transplant atherosclerosis.
Clean ISR & WO by International Search Authority
Entered National Phase: US, Canada, Europe, Australia, Japan, China (Oct 9th, 2017)
Will provide COM protection for the API/ VSR LNP until at least 2042
US2020/041957 – US20 841 595.0 - EU 20 841 595.0
“METHODS AND COMPOSITIONS FOR TREATING PULMONARY ARTERIAL HYPERTENSION”
US 17/721,666 – US 16/086,809 “METHODS AND COMPOSITIONS FOR TREATING ATHEROSCLEROSIS”
US 9,238,716 B2 - WO 2012/135025 A2 “Conjugated Lipomers and Uses Thereof”





